The Synthesis of Complex and Diverse Compounds from Natural Products
The majority of commercially available compound screening collections consist of simple, planar compounds that lack significant stereochemistry. While such compounds are suitable to modulate certain biological targets, they are generally not effective against more complex targets (e.g., protein-protein interactions, protein-DNA interactions). In addition, the majority of FDA approved anticancer and antibacterial agents are complex natural products, compound with physiochemical properties vastly different from those in compound screening collections. We have initiated a program whose goal is to create complex and diverse compounds. To do this, we take readily available natural products, and in ~5 or fewer chemical steps change them to compounds that are complex, but diverse from the parent natural product and from each other.
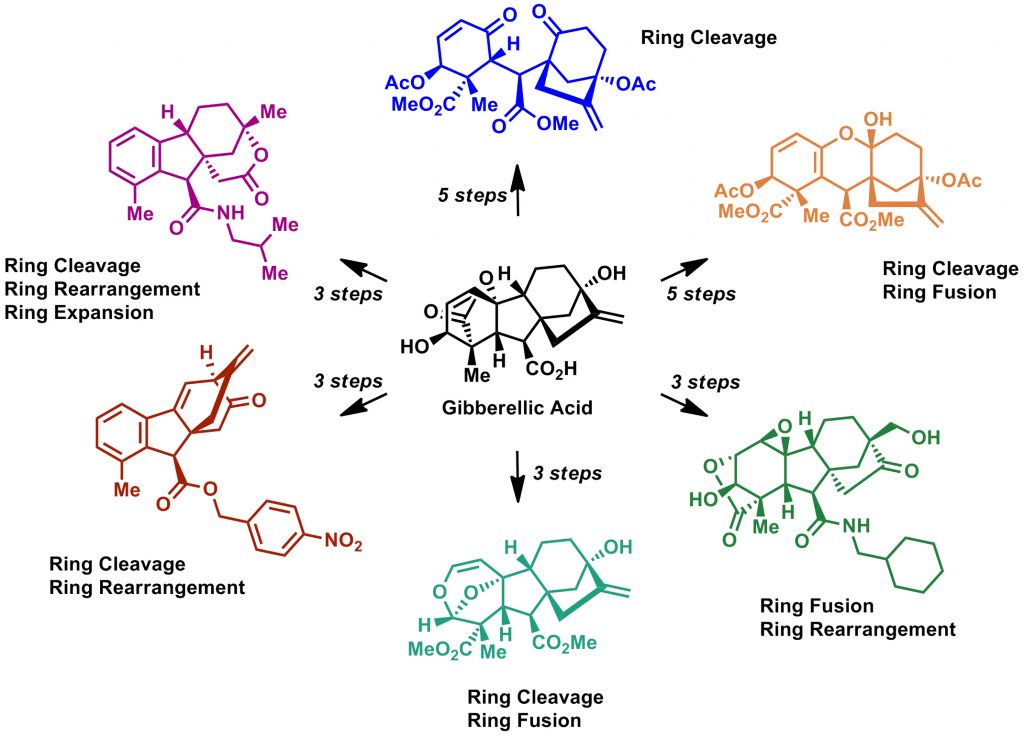
Gibberellic acid was rapidly converted into six highly diverse and complex scaffolds.
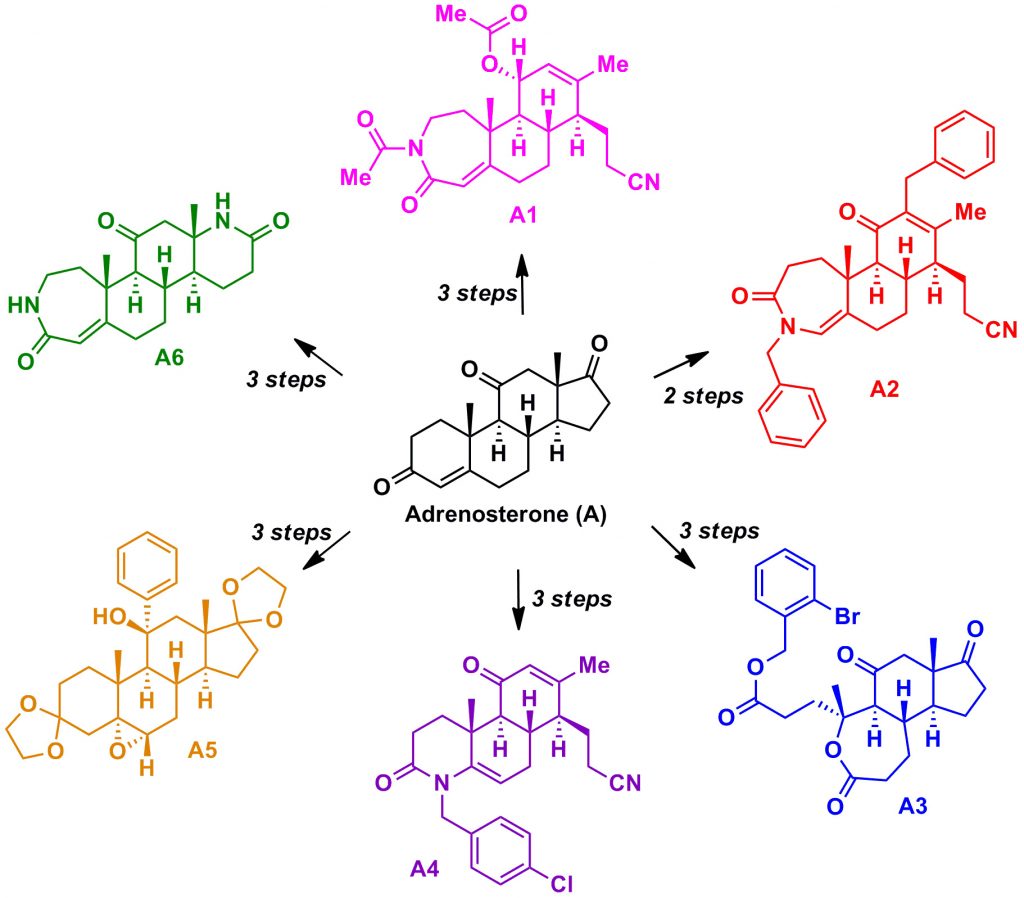
Ring distortion afforded many complex structures starting from Adrenosterone.
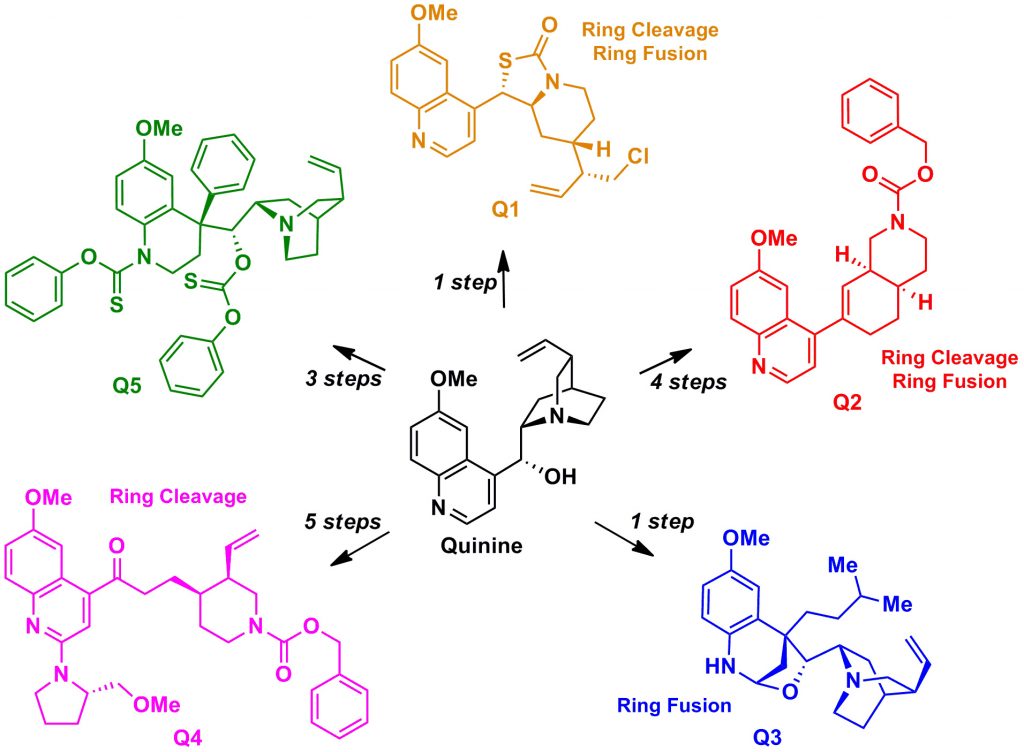
Ring distortion afforded many complex structures starting from Quinine.
Related Publications:
- A catalytic process enables efficient and programmable access to precisely altered indole alkaloid scaffolds
Huang Y, Li X, Mai BK, Tonogai EJ, Smith AJ, Hergenrother PJ, Liu P, Hoveyda, AH
Nature Chem. 2024, published on web February 19, 2024.
Link to journal
- Limonin as a Starting Point for the Construction of Compounds with High Scaffold Diversity
Furiassi, L; Tonogai, E.J.; Hergenrother, P.J.
Angew. Chem. Int. Ed. 2021, 60, 16119-16128
Link to journal
Article PDF
- Re-engineering natural products to engage new biological targets
Motika, S. E. and Hergenrother, P. J.
Nat. Prod. Rep. 2020, 37, 1395-1403.
Link to journal
Article PDF
- Diverse compounds from pleuromutilin lead to a thioredoxin inhibitor and inducer of ferroptosis
Llabani, E.; Hicklin, R. W.; Lee, H.-Y.; Motika, S. E.; Crawford, L. A.; Weerapana, E., Hergenrother, P. J.
Nature Chem. 2019, 11, 521
Link to journal
Article PDF
Nature Chem. highlight
- Preparation of structurally diverse compounds from the natural product lycorine
Tasker, S. Z.; Cowfer, A. E.; Hergenrother P. J.
Org. Lett. 2018, 20, 5894-5898.
Link to journal
Article PDF
- Access to a Structurally Complex Compound Collection via Ring Distortion of the Alkaloid Sinomenine
Garcia, A.; Drown, B. S.; Hergenrother, P. J.
Org. Lett. 2016, 18, 4852-4855.
Link to journal
Article PDF
- Synthesis of Bridged Oxafenestranes from Pleuromutilin
Hicklin, R. W.; López Silva, T. L.; Hergenrother, P. J.
Angew. Chem. Int. Ed. 2014, 53, 9880-9883.
Link to journal
Article PDF
- Synthesis of Complex and Diverse Compounds through Ring Distortion of Abietic Acid
Rafferty, R. J.; Hicklin, R. W.; Maloof, K. A.; Hergenrother, P. J.
Angew. Chem. Int. Ed. 2014, 53, 220-224.
Link to journal
Article PDF
- Natural Products as Starting Points for the Synthesis of Complex and Diverse Compounds
Morrison, K. C.; Hergenrother, P. J.
Nat. Prod. Rep. 2014, 31, 6-14.
Link to journal
Article PDF
- A Ring Distortion Strategy to Construct Stereochemically Complex and Structurally Diverse Compounds from Natural Products
Huigens, R. W.; Morrison, K. C.; Hicklin, R. W.; Flood, T. A.; Richter, M. F.; Hergenrother, P. J.
Nature Chem. 2013, 5, 195-202.
Link to journal
Article PDF
C&E News Highlight
Faculty of 1000 Highlight
Nature Chemistry News
YouTube Animation