How Fuel Cells Work
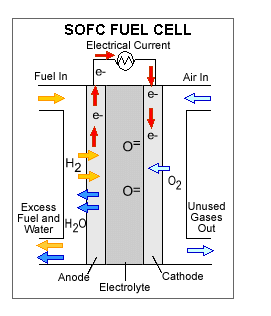
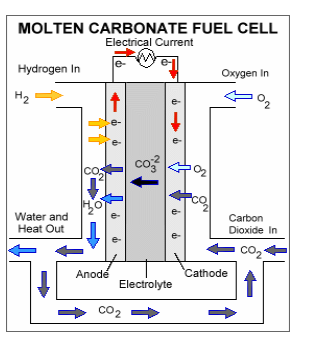
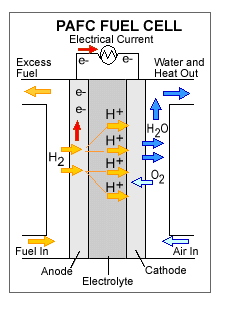
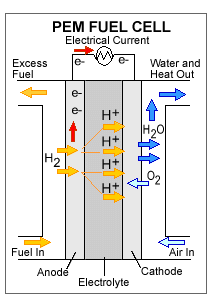
1st –> Hydrogen fuel enters the cell at the anode, which is positively charged but negatively biased.
2nd –>Here, the Hydrogen splits and forms positively charged ions and negatively charged electrons.
3rd –>The Hydrogen elctrons are not able to go directly to the cathode, which is negatively charged, but positively biased. The electrons have to take a separate path to the cathode and this is how the electrical current is produced. The Hydrogen ions, however, are able to go directly to the cathode.
4th–> Electricity is produced and the Hydrogen ions combine with Oxygen and form a by-product of water and leaves the cell.
The picture below shows a PEMFC , used in automobiles